
Biology
Chapter 2: Atoms and Molecules

Lecture
The Chemical Basis of Life
Web Lecture
- Holistic and Reductionist approaches
- Levels of Organization and Emergent Properties
- Atoms
- Chemical Reactions
Holistic and Reductionist Approaches
There are lots of ways to investigate phenomena. Two important ways are to look at a thing and break it down into smaller and smaller parts, and to look at a whole bunch of things all at once as a single system. As we go through the material in biology this year, we'll move from the analytic break-it-down method to the synthetic see-it-all-at-once method. Both are necessary and important ways to help us understand what life is, and how living organisms interact.
During the first part of this course, we use the reductionist method to break living organisms into their smallest common "pieces" -- atoms, molecules, cell components, and cells. The cell is the common component of everything we have defined as living. If we understand something about how cells work, we understand something about the way every living thing on earth organizes its own living matter and how it uses energy to stay alive, grow, and reproduce.
At the other end of the methodological approach spectrum is the holistic approach. This approach tries to see everything at once as part of an interconnected system. If you've ever stood at the edge of the Grand Canyon at sunset, or on the meadows at Paradise on Mount Rainier, or at the ocean shore, you have some sense of why a holistic approach is both necessary and difficult. Words and descriptions can't convey the size and complexity and wonder of the natural phenomenon in front of you in these places.
We realize when we study the structure of cells that we see only part of what cells are; the other pieces is how cells relate to one another. As we move beyond our study of cells by themselves, we'll take up cell reproduction and inheritance, then the formation of cells into tissues, organs, organ systems, and organisms. At the end of our studies, we'll use a more holistic approach to put organisms into populations, communities, and ecosystems, and study the interaction of living things.
Levels of Organization and Emergent Properties
As we move from the most common of characteristics of matter, atoms, to molecules, we start separating out those molecules which we don't tend to find in living things from those which are the essential building blocks of cells. As we look at cells, we start to make distinctions between all the characteristics that are common to all cells, and the characteristics that are different in different kinds of cells. All cells have some kind of cell barrier that defines the boundary of the cell, but the cell walls in bacteria and archaea different between the two groups of single-celled prokaryotic organisms, and differ even more from the Protists, the single-celled eukaryotic organisms.
With each level of higher organization, we discover different, new characteristics that emerge as part of the common properties of that level of organization. Different organisms express these characteristics in different ways. Where properties are common, we can look for patterns that allow us to classify things and make predictions about behavior. But everywhere we look in the living world, we find exceptions to our classifications and predictions, reminding us that biology is not an exact science in the same sense as physics and chemistry.
Atoms
The art of drawing conclusions from experiments and observations consists in evaluating probabilities and in estimating whether they are sufficiently great or numerous enough to constitute proofs. This kind of calculation is more complicated and more difficult than it is commonly thought to be.
Antoine Lavoisier
While Democritus and Leucippus proposed around 450 B.C. that all matter was made of bits that could not be broken down into smaller components (atomi = "without a cut": think of "lobotomy" which means cutting into the head!), there was no observational evidence to support their claim. Empedocles' theory of four elements, earth, fire, water, and air, dominated the study of matter for nearly two millennia until attempts to explain the behavior of matter using just these elements broke down in the seventeenth century. In the eighteenth, the chemist Lavoisier studied the behavior of gases with greater accuracy than any of his predecessors. He determined that there were at least 32 elements, and very likely many more, setting off a hunt for elements that consumed many chemists and physicists.

Calorimeter similar to that used by Lavoisier at the Musée des Arts et Métiers, Paris.
Photo © 2009 Christe Ann McMenomy
In the next generation, John Dalton in England began to realize that there were two ways to measure combinations, by weight and by volume, and in both measures, Lavoisier's new elements combined in whole number ratios. His discoveries led to a recovery of the theory of atomic matter, but Dalton thought atoms were still indivisible, the smallest possible building blocks of matter.
| Parts Hydrogen by weight | Parts Oxygen by weight | Ratio of water by weight |
| 2 | 16 | 1:8 |
| Parts Hydrogen by volume | Parts Oxygen by volume | Ratio of water by volume |
| 2 | 1 | 2:1 |
It was Avogadro's hypothesis (originally rejected for six decades) that volume and numbers of particles were related that eventually led to the realization that water was 2 atoms of hydrogen to 1 atom of oxygen: H2O.
Over the next century, chemists discovered that 92 elements occurred in nature, and that atoms were actually divisible, composed of even more fundamental and very tiny particles that move constantly and take up a lot of space:
- protons, positively-charged massive particles in the nucleus or center of the atom
- neutrons, uncharged massive particles also found in the nucleus
- electrons, negatively-charged particles that are very light by comparison to protons and neutrons, and in constant motion in the space around the nucleus.
It is the electrical charges which hold the atom together — and hold atoms in combinations of molecules.
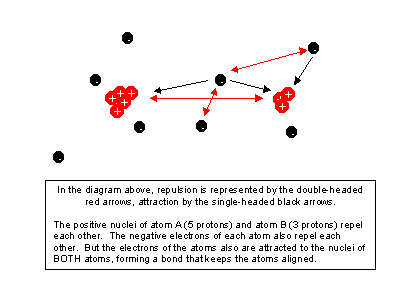
Basic Atomic Properties and Reactions
All of the reactions that support life are based on chemical reactions between atoms: interactions where atoms rearrange themselves into different molecules, releasing or consuming energy as they change organization.
Click on the image of the two atoms to review basic properties of atoms and their interactions.
Atoms in molecules are held together by the electrical attraction of their own positive nuclei for the electrons of other atoms. A chemical bond is a particular balance between attractive and repulsive forces in a specific arrangement of atoms. If we move the atoms, we change that arrangement, and we can create new arrangements (new molecules). Such a rearrangement takes work to pull the atoms out of their current state, just as lifting a book to a higher shelf requires work. Putting them in a new arrangement, though, may actually release energy, so there are different kinds of chemical reactions.
- Exothermic, or exergonic reactions release energy.
- Endothermic, or endergonic reactions absorb energy.
Most metabolic reactions are endothermic at normal body temperatures, so they require help in the form of catalysts to get started, and they are often coupled with an exergonic reaction that provides any extra needed energy to fuel the endergonic reaction. When we do an analysis of a metabolic process, we have to add up the energy requirements of all of the steps involved to see the "overall" energy absorption or output. Keep this in mind when we get to the processes of photosynthesis and cellular respiration.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.
