
Biology
Chapter 5: Energy and Enzymes

Lecture
Cell Structure and Organelles
Web Lecture
- Concepts of Energy
- Enzymes
Energy Basics
Mass, Energy, and Work
We are concerned now with how the cell acquires the energy, how it stores it, and how it frees energy from storage for use. So we need to establish some very basic concepts about force, energy, and matter.
Matter is anything that has mass and takes up space. Mass isn't quite the same thing as weight; mass is a measure of the amount of matter and doesn't change when an object moves from one gravitational field to another. If we transport a jar of jelly beans weighing one pound from the earth to the moon, it still has the same mass and the same number of sugar atoms, but it will only weight 1/6 pound because the moon's gravitational pull (force) is 1/6 of the earth's. Weight is a measure of the force exerted on the jelly beans by the gravitation pull of the nearest planetary-type body.
Energy is a measure of the work required to change the direction of motion or the state of matter. One way to calculate work is to look at the force required to move an object a given distance. To move the jelly beans three feet across the table and out of the reach of your little brother is 1 pound * 3 feet, or 3 foot-pounds:
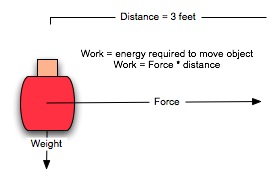
Of course, this is oversimplified: you need to expend an excess of 3 foot-pounds worth of energy, because you are also moving your arm and part of your body, lifting the jar against the earth's gravitational field, and utilizing hundreds of chemical reactions requiring small amounts of energy at the cellular level, just to free the energy you actually use to move the jar.
We used to measure such organic expenditures in terms of the equivalent amount of heat energy. This was the standard for many years, because all forms of energy—mechanical energy, chemical energy, electrical energy, potential energy—can all be converted to heat energy. The reverse energy flow from heat to another form of energy is possible only when heat flows from a hotter area (heat source) to a colder area (heat sink); under this condition, it can be converted into mechanical energy and do work. In biological systems, the flow is from the light energy of the sun into chemical bond energy (food created by producers) to chemical bond energy (food stored by consumers) to chemical bond energy (chemicals broken down by decomposers).
Until recently, the standard measure of energy was the calorie. One calorie is the amount of heat necessary to raise a cubic centimeter (or milliliter) of water 1 degree Centigrade from 20 degrees centigrade at sea level. This unit was chosen because it was easily calibrated most places on the earth, with just a volume measure of a readily available substance—water. Other forms of energy were calculated as the equivalent amount of heat energy and expressed in calories. In the last decade, however, biologists have chosen to use another unit, the kilojoule, because cells do not actually use heat energy to do work. [Cells have to maintain a narrow temperature range—part of their tendency to homeostasis, so heat flows from high temperature areas to low temperature areas within the cell are extremely rare.] A joule is equal to 1/4.184 calories, or .239 calories; or one kilogram-meter2/sec2.
By the way, the calories in your soda pop are really Calories (capital-C); nutritionists use that term as shorthand for a kilocalorie.
Every object has a certain amount of energy associated with it, simply because of its composition and position relative to other masses. This potential energy is the energy available for the object to do work. If you pick your book up off the floor and set it on the table, you store energy in it, equivalent to the distance you moved it against the force of the earth's gravitational field. When we add salt to the solution around our cucumber slices, we store energy in the ordered difference of the concentrations (salt water to cucumber cell contents). When your body combines two glucose molecules together to make sucrose, it stores energy in the chemical bond between the two glucose molecules. All these forms of stored energy are potential energy.
The energy stored in the book on the table, in the salt solution, and in the sugar bond can be recovered. If the book falls from the table, the potential energy released by its change is position is converted into sound energy and heat energy. Of course, if you are clever, you can attach your falling book to something you want raised from the floor through a system of strings and pulleys, and do useful work instead of merely creating a disturbance. Your cells are very thrifty; they can take advantage of energy stored in the difference in concentration and that in the chemical bonds of fats and carbohydrates. When osmotic pressure carries water out of the cucumber cells, down the sodium concentration gradient, and into the solution, the cell membranes can do work and pump other compounds into the cell, against the concentration gradient for that compound. When the chemical bonds in the sucrose molecule are broken apart, the energy is used to drive other metabolic chemical reactions which promote growth, or movement, or some other useful function.
The Laws of Thermodynamics
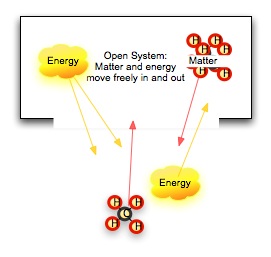 |
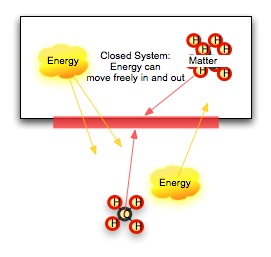
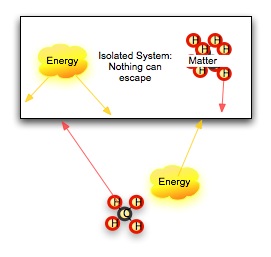
No conversion of energy is completely efficient, however. Our clever book-elevator loses some heat energy to friction in the pulley system no matter how carefully we build it. Our cells maintain a constant temperature, and our bodies lose heat to our surroundings. What is happening? Let's first look at what we mean by system. A system is a group of objects that lie within some defined boundary. The boundary can be real (a wall, a thermos jar) or it can be imagined (the edges of a hydrogen-II cloud in space). In an open system, both energy and matter can cross this boundary. In a closed system, matter cannot cross the boundary, but energy still can. In an isolated system, nothing crosses the boundary. What happens in an isolated system does not -- it cannot -- affect the surrounding universe. The First Law of Thermodynamics stipulates that the total energy of an isolated system cannot change. Another way to say this is that energy is not created or destroyed. We think the universe is a isolated system, so the total energy of the universe does not change with time. The Second Law of Thermodynamics stipulates that the total disorder of a isolated system, measured by the amount of useless heat energy (energy which cannot do work) is increasing. This does not contradict the first law. There are two kinds of energy here: one in several forms which we can use to do work, and one random, disordered form of energy which does no work. |
 |
|
 |
Any time a process produces more order in a local area, disorder increases elsewhere. When you pick up your room, you use energy which is converted into heat, and radiated away from your body, away from your room, away from earth, and out into space. The total energy of the universe (your room, your body, the earth and all space) does not change. Your room is neater (and your parents happier), but the universe is a little more run down as a result—it has less useful energy than it did before.
The total potential energy of the system, or enthalpy, exists as free energy (energy available to do useful work) and as entropy, or useless energy. We can write this as a simple mathematical sum:
G = H -TS, or free energy = enthalpy - temperature * entropy
If a process is 100% efficient, there is no increase in entropy, and the free energy available to do work is equal to the total potential energy. But no process is 100% efficient. In a isolated system, enthalpy is constant, and entropy increases, so during the process, the amount of free energy decreases. If the system is open and we have a fixed amount of free energy (G) to do work, and we create an increase in local order (S decreases), then H must also decrease, that is, some energy must escape to the outside and increase the entropy outside the local system, to balance the equation.
Metabolic reactions
Cells cannot use heat flows to produce energy, because they cannot support the differences in temperature this would require. Instead, they depend on many thousands of small-scale, interrelated chemical reactions to provide energy for their metabolic functions (growth, digestion, waste elimination, reproduction, repair). This system of reactions is sometimes referred to as a metabolic web. Reactions which synthesize complex molecules (like making sucrose from glucose or proteins from amino acids) are anabolic. Reactions which break down complex molecules into their simpler components are catabolic.
Reactions occur because there is a change in the energy state. We can write the general case for energy change during a reaction as
ΔG = ΔH - T * ΔS
where the deltas indicate "change in". Read this as "the change in free energy during the reaction is equal to the change in the total reaction or enthalpy minus the temperature * the change in entropy".
We used to talk about exothermic "heat-producing" and endothermic "heat-absorbing" reactions, but we can get a more accurate picture of the energy changes involved in a reaction from looking at the free energy exchange. In some reactions, the products contain more usable energy than the reactants, that is, there is a gain in free energy: ΔG is positive, the reaction is endergonic. These reactions require a constant input of energy to start and to continue. In others, the products contain less energy than the reactants: there is a decrease in free energy: ΔG is negative, and the reaction is exergonic. While these reactions may require energy to start running (activation energy), once started they will continue to run on their own energy output as long as reactants are available. We call such reactions spontaneous, but the term doesn't mean that the reaction occurs all at once. Usually, exergonic reactions are exothermic and spontaneous, but some endothermic reactions, like ice melting, are also spontaneous.
Some reactions are reversible. The difference in free energy between the reactants and the products is so small that minor changes in the environment of the reaction, such as a temperature change, or an increase in the concentration of one of the compounds involved, is enough to cause the reaction to "run in the other direction" (this is called Le Chatelier's principle after the French scientist who first defined it). Actually, the reaction is running both ways all the time, if viewed at the individual molecule level. The net direction of the reaction is in the direction of increasing concentrations. The net effect of the reaction is to bring the concentrations back into equilibrium. A reaction is at equilibrium when the concentrations of the participating compounds are no longer changing. Such a system can do no work, because nothing (taken as a whole) is changing.
We can write the relationship of concentrations in a chemical reaction
A + B → C + D
as
K = [C] * [D] / [A] * [B],
where K is a constant for the specific reaction, and the brackets indicate we are looking at concentrations of the compounds (usually in solution or gases). If the concentration of C is increased, the reaction will run in a direction which changes the concentrations of D, A, and B so that K remains unchanged.
Cells are almost never at equilibrium. Instead, they control the concentrations of their components using exergonic, endergonic, and reversible reactions, depending on the availability of products and reactants and the current task to be accomplished. Sometimes a combination of exergonic and endergonic reactions are required to create a specific compound product. If the reaction A→B is endergonic and B→C is exergonic, the reaction may proceed if the energy released by the second reaction is greater (to account for inefficient energy conversion) than that required by the first.
That is why we envision the set of reactions as a web: they are really a complex set of interrelated processes, and modifying one reaction will have a cascade effect through a number of other reactions.
Energy Currency
The cells of our bodies store energy easily and efficiently in the chemical bonds of particular compounds. The most important of these are adenosine triphosphate and nicotinamide adenosine dinucleotide. [Chant these in front of the mirror until you can say them easily, then you will remember what ATP and NAD+ are when you run into them in your reading: add-DEN-oh-zeen try-FOS-fate, NICK-oh-teen-AM-ide die-NUKE-lee-oh-tide].
Think of ATP as a set of pop beads. One of the beads is adenosine, and it is glued to another bead, which represents a 5-carbon sugar called a ribose. The sugar is glued to a phosphate ion. This phosphate ion has a pop-bead hole, into which you can plug another phosphate ion, which itself can take one more pop-bead phosphate.
When you pull the pop-beads apart, the popping noise represents the energy released. When you put them together, the push you must exert to join them is the energy you are storing in the bond. Pull one phosphate off of ATP, and you have a two-phosphate molecule left: adenosine DIphosphate. Pull a second phosphate off, and you have a one-phosphate molecule left: adenosine MONOphosphate. The cell combines the phosphate ions with other molecules, usually in an exergonic reaction, which releases more energy. Cells are constantly creating ATP and breaking it down for its energy content; they do not use it as long-term energy storage.
The "tri"-phosphate ion group breaks off at the P-O bond, releasing energy.
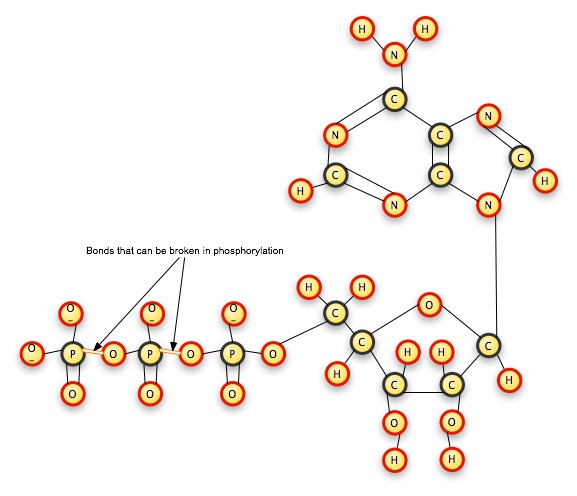
Adenosine Triphosphate -- the adenosine group is in the upper right (two nitrogen-carbon rings), the ribose on the lower right, and the phosphate chain to the left.
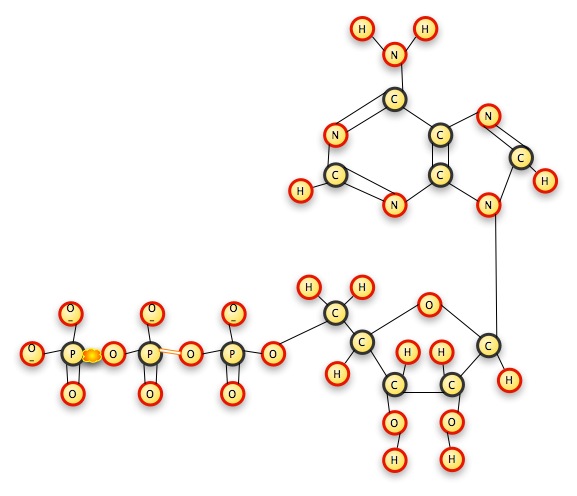
The "tri"-phosphate ion group breaks off at the P-O bond, releasing energy.
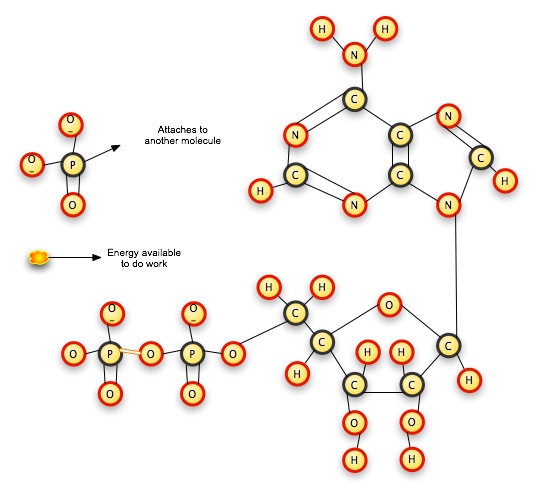
The isolated phosphate ion quickly combines with another molecule somewhere, and the released energy is used by the cell to do work. The remaining molecule, now ADP (adenosine diphosphate) is available to release further energy by giving up another phosphate group (and becoming AMP), or to store more energy by taking on another phosphate group (and becoming ATP again).
Another method of energy storage and transfer involves passing around electrons. Remember our discussion of oxidation/reduction reactions in chapter 2? In a redox reaction, one substance loses electrons (oxidation) while another gains electrons (its ability to combine further is reduced). In biological redox reactions, the hydrogen atom is usually removed in its entirety from the molecule to which it is attached by a covalent bond; ¨the energy represented by the electron bond goes with the hydrogen atom. NAD+ will accept a hydrogen atom and extra electron to become NADH; the bond represented by the two electrons is a form of energy storage.
Enzymes
Cells need to perform numerous chemical reactions, not all of which are exergonic. Assume we have a chemical reaction: A + B → C + D, with a high activation energy. The reaction cannot proceed without pumping a lot of energy into it. Now into the solution of A and B, we introduce a new compound E. Suddenly the reaction takes off, and after some time, we have lots of C and D, and very little A and B, but the amount of E doesn't change.
E is a catalyst. The reaction which wouldn't occur otherwise, takes place easily in its presence. It remains unchanged throughout the reaction process and continues to act as a catalyst, facilitating the reaction until all the reactants are used up.
Your body has hundreds of different kinds of biological catalysts, called enzymes. They work by forming temporary compounds with the substrates (reactants) of a chemical reaction. The substrates fit into or attach to the enzyme at a particular place, called the active site. The enzyme can simply hold the substrates close enough to interact and form a new compound (lock and key model), or it may actually rotate the substrates into an appropriate orientation (induced fit model). Enzymes are usually very efficient at what they do; a few enzymes may facilitate millions of reactions in a short period of time. Enzymes are also specific: a single enzyme usually facilitates a single reaction (although some enzymes work with classes of reactants). That is why you need so many different kinds.
Coenzymes
Most enzymes are proteins. Some enzymes have a protein component (the adenosine) and another chemical compound component (the cofactor). Because the protein and the cofactor are produced in different parts of the cell and neither alone is a catalyst, only when they come together do they actually work as an enzyme. When cofactors are made of non-polypeptide compounds, they are called coenzymes. Many vitamins work as coenzymes, which is why you need them so badly: Without them, specific important chemical reactions cannot take place, and your cells become ill, inefficient, or die.
As with any chemical reaction, enzyme-assisted reactions are affected by the temperature. Increasing temperature increases collisions between molecules, making it possible for more reactions to occur in a shorter period of time. It can also cause the enzyme to break apart. Most enzymes have a small range (35-40 degrees centigrade) within which they can act as catalysts. Human body temperature, which you commonly know as 98.6° Fahrenheit, works out to 37° centigrade, so most metabolic reactions occur in the range where enzymes are most effective. If your body temperature drops below 93°F (33.85°C) or rises above 105°F (40.85°C), enzymes cease to function effectively, and your metabolic processes slow down to a rate which may be life-threatening.
Enzyme activity is also affected by acidity. Remember that acids and bases dissolved in water cause an excess of H+ and OH- ions, respectively. Since enzymes are proteins made up of amino acids, many of which are polar, increasing the number of charged particles in the enzyme's environment will affect the ability of the enzyme to orient itself; it may even cause the enzyme to change shape, which in turn will affect the ability of the active site to attach the substrate molecules.
Because enzymes and enzyme activity are easily affected by changes in the environment, a cell can control enzyme activity by altering its environment or by altering the enzyme. Increasing acidity or alkalinity is one possibility. Using a chemical inhibitor, which attaches to the enzyme in place of the target substrate (competitive inhibition) or attaches elsewhere and changes the shape of the enzyme so that it cannot attach to the target substrate at all (allosteric regulation), is another way for the cell to prevent enzymes from assisting a reaction. Some inhibitors can attach to the enzyme only temporarily (reversible inhibition); when they leave, the enzyme is able to continue with its job of making some chemical reactions easier. Other inhibitors permanently alter or even destroy the enzyme (irreversible inhibition). We call most such inhibitors poisons.
By the way, the word model should be a clue that we do not entirely understand how enzymes work. A model is an imitation (some people prefer the term analogy) of reality. Scientists are constantly tinkering with the model, trying to make it more accurate. The fact that we have two proposed models of enzyme activity indicates that we aren't sure which is right. In this case, the models are not mutually exclusive: both code be correct if applied to different enzymes, or to the same enzyme in different circumstances.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.