
Physics
Chapter 27: 7-13 Early Quantum Mechanics

Web Lecture
The Bohr Atom
Introduction
When I began to consider these difficulties I was chiefly struck by facts. On the one hand the Quantum Theory of Light cannot be considered satisfactory, since it defines the energy of a light-corpuscle by the equation W = hν, containing the frequency ν. Now a purely corpuscular theory contains nothing that enables us to define a frequency; for this reason alone, therefore, we are compelled, in the case of Light, to introduce idea of a corpuscle and that of periodicity simultaneously.
On the other hand, determination of the stable motion of electrons in the atom introduces integers; and up to this point the only phenomena involving integers in Physics were those of interference and of normal modes of vibration. This fact suggested to me the idea that electrons too could not be regarded simply as corpuscles, but that periodicity must be assigned to them also.
In this way, then, I obtained the following general idea, in accordance with which I pursued my investigations:--that it is necessary in the case of Matter, as well as of radiation generally and of Light in particular, to introduce the idea of the corpuscle and of the wave simultaneously: or in other words, in the one case as well as in the other, we must assume existence of corpuscles accompanied by waves. But corpuscles and waves cannot be independent of each other: in Bohr's terms, they are two complementary aspects of Reality: and it must consequently be possible to establish a certain parallelism between the motion of a corpuscle am propagation of its associated wave. The first object at which to aim, therefore, was to establish the existence of this parallelism.
Louis de Broglie — Nobel Prize acceptance speech, 1929
Outline
The Bohr Atom
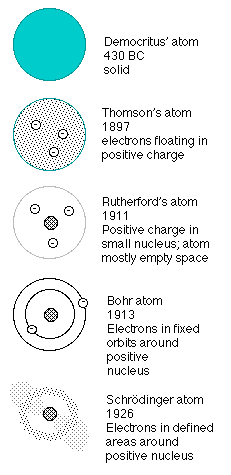
The first proposal that matter was made of indivisible small particles comes from the Greek philosophers Democritus and Leucippus. Their theories did not gain acceptance at the time, since no evidence existed to support them. Robert Boyle returned to this theory in the seventeenth century when he proposed a corpuscular matter theory in The Skeptical Chemist. John Dalton's theory of chemistry rested firmly on the idea of atoms of different types that could combine as discrete units, supported by the whole-number ratios of elements found in molecular combinations such as water (2 volumes of hydrogen to 1 volume of oxygen, or 2 mass units of hydrogen to 16 mass units of oxygen). By the mid-nineteenth century, the idea of the indivisible unitary atom was firmly entrenched in chemical and physical theory, only to be challenged by the radioactivity discoveries of the Curies and the new "rays" emitted in cathode tubes, especially beta rays, which has a mass that was only a very small fraction (about 1/2000) of the hydrogen atom.
With the investigations of the early twentieth century, the model used to describe the atom changed drastically. Thomson's "plum pudding" model in which small negative particles moved sluggishly in fluid plastic mass of positive charge failed to account for the results of Rutherford's experiment, which supported an atom with a concentrated positive nucleus surrounded by an electron fog. However, such a model could not explain either the formation of molecular bonds or observations of absorption and emission spectra.
Bohr's atomic theory combined quantum theory with Rutherford's observations, and accounted for spectra by assuming that electrons could absorb and emit only specific quanta of energy. It wasn't until de Broglie applied the principle of wave nature to electrons that physicists could justify this assumption, however. Further exploration of the wave nature of matter and the Heisenberg uncertainty principle modified the view of the atom still further: it was not possible to confine or locate electrons precisely, but only to describe the probability that they existed in a given volume, based on their energy level.
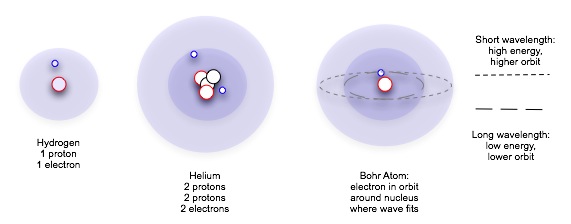
Study the wave nature of matter at the University of Colorado Bose-Einstein Condensate website[requires JAVA]. You may wish to explore other pages on this site as well.
Practice with the Concepts
Discussion Points
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.