
Biology
Chapter 5: Cell Membranes and Energy

Lecture
Transport Across Cell Membranes
Web Lecture
- Membrane Composition
- Proteins
- Transport
- Receptor Mediated Transport
- Carrier Mediated Transport
- Exocytosis and Endocytosis
Passive and Active Transport
Membranes
Biological membranes are special surfaces which separate one part of a cell from another, or whole cells from each other. You can think of them as a kind of sealable trash bag, which stretches a little and reshapes itself to hold whatever it contains.
Membranes are made mostly of certain lipids (fat molecules), along with some proteins and carbohydrates. Phospholipid molecules (phosphorylated alcohol + glycerol + 2 fatty acids) line up in long double rows called a lipid bilayer to form membranes for cells and for some organelles (like the nucleus and mitochondria). The fatty acid chains of hydrocarbons are not uniform. Some carbon atoms bond to only one hydrogen, and must double-bond with another carbon in a similar state. This causes the fatty acid tails to kink, and puts gaps in the bilayer. So it isn't a nice, uniform surface, even if we don't take into account all the proteins, carbohydrates which are embedded in it.
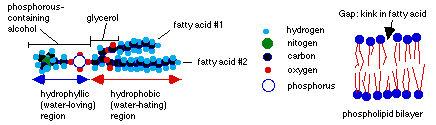
The individual phospholipids are attracted to one another by Van der Waals forces. Because these forces are so weak, the phospholipid molecules can not only rotate in place, they can also move from place to place, so the bilayer is a fluid. Decreasing the temperature causes it to form a gel (semi-solid); increasing the temperature causes it to weaken and even break apart. Cholesterol molecules embedded in the bilayer act as a kind of temperature buffer: if the temperature increases too much, they dampen the movement of the phospholipid molecules so the cell wall doesn't disintegrate. If the temperature decreases, they hold the phospholipid molecules apart, so they don't form a gel.
Proteins
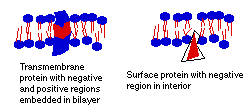
Peripheral proteins are found on the surface of the membrane, where they are held by non-covalent bonds to proteins embedded in the membrane. The embedded or integral proteins may actually cross the entire membrane, since it is usually about 10 nm (that's 10 * 10-9 meters) thick, and most proteins are larger than 10nm. These are called transmembrane proteins. Both transmembrane and peripheral proteins have areas which are charged because of the polar amino acids they contain; and these charged areas must line up with the polar and water-loving areas of the phospholipid molecules.
Proteins are not evenly distributed on the cell membrane. There are generally fewer proteins in the P side (interior) than on the E side (exterior). This gives the inside a "smooth" look and the outside a "rough" look.
Proteins embedded in the membrane have a number of functions, including transporting molecules and information across the membrane, catalyzing (aiding) cell reactions, and binding cells together.
Transport
Because the membrane separates the interior of the cell from its environment, it also control what gets into and out of the cell. Cells can therefore regulate their own composition, which is a very important requirement for their survival.
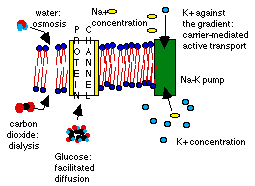
Selective permeability is the ability of a membrane to allow some molecules to pass through but not others. The kinks in the fatty acids leave gaps in the bilayer through which water molecules, small gases like CO2, small polar and some larger non-polar molecules can pass easily. Larger polar molecules and ions must use special mechanisms to cross the membrane.
Diffusion is the net movement of particles between two areas of different concentrations such that the higher concentration is diffused, or lowered, and the lower concentration raised. Individual particles of a substance may move into an area of higher concentration of that substance, but over time, more molecules of the substance will move to areas of lower concentration than to the higher concentration areas. We call the difference in concentrations a concentration gradient. A gradient is just a slope: in math, it is the slope of the line. On the surface of the earth, it is a hill. We can talk about the temperature gradients you would observe as you walk from a sunny area into the shade.
In passive transport, the net flow of energy is always from a high point to a low point, "down the gradient". Warm air cools down by warming up the colder air around it. Concentrations become diffuse by moving their solutes (the thing which is dissolved in the solution) into areas of less concentration. The rate of flow of particles through a membrane by diffusion is a function of the solute size, charge on the solute (if the membrane is made of charged or polar particles, and cell membranes are), and the temperature, since the hotter it is, the faster particles will move.
In passive transport, the net result is always the dilution of the higher concentration. In dialysis, the solute particles move across the membrane to the area of less concentration. In osmosis, the solvent (what the solute particles are dissolved in, usually water) moves across the membrane from an area of lower concentration (hypotonic solution) into the area of higher concentration (hypertonic solution). The pressure exerted by this movement of water is osmotic pressure. If the osmotic pressure drives water from the cell, the cell collapses in a process called plasmolysis, and may even die. Plant and fungi cell walls, and some single celled animals as well, resist water loss by controlling their internal concentrations so that water flows into the cell until it exerts enough turgor pressure to make the cell rigid, like a water balloon.
Receptor Mediated Endocytosis
Some large molecules (including viruses) can be passed into the cell through endocytosis. In this process, a receptor protein on the cell membrane binds to the molecule, which triggers other proteins inside the cell, usually a form of clathrin, to also bind to the membrane. As a result, the membrane begins to surround the molecule, forming a cap over the outside of the molecule, and a patch on the existing membrane that will eventually dissipate and open, dropping the molecule and the receptor protein into the cell interior.
Carrier Mediated Transport
Polar molecules can't cross the cell membrane by dialysis, because the localized charges on their surface are repelled by localized charges on the phospholipids. In facilitated diffusion, protein "gates" allow ions and large polar molecules to flow down the concentration gradient. This is like opening a larger hole in a strainer: the particle would normally go that way anyway, but it is too big to get through the regular strainer holes.
Restricting the flow of ions and polar molecules this way is actually a good thing, normally, because the cell can't afford to lose its store of nutrients to the less-concentrated fluids outside the cell. It does need a way to pull molecules across the membrane against the concentration gradient, so that it can get more nutrients even when the outside solution is less concentrated that its own interior.
A set of proteins in the membrane provide carrier-mediated active (uphill against the gradient) transport. These use energy to "pump" ions and molecules into (or out of) the cell against the gradient curve. The sodium-potassium pump has been well researched in the last two decades, since it was identified as the source of chlorine imbalance in the cells of patients with cystic fibrosis. I'm not going to describe it here; we will look at it in detail in a later chapter.
Some pumps use a flow of one particle down the gradient curve to help fuel pushing another particle up the gradient curve. This is a co-transport system, and it is more energy efficient than a simple active carrier-mediated protein gate, since it gets back some of the energy it expends pushing one molecule up the concentration gradient from the down-hill flow of the other molecule, and it uses one mechanism to facilitate the flow of two particles.
Some molecules require multiple mechanisms to move across different membranes and reach their target. Glucose moves into the stomach lining cells through a sodium driven co-transport system (the sodium moves down the gradient curve by diffusion), then moves out of the cell into the blood vessels through a transporter mediated diffusion process. The sodium concentration in the stomach lining cell is regulated through a sodium-potassium pump, which moves excess sodium out of the cell on one side, so that sodium will continue to diffuse into the cell on the other, bringing glucose with it.
All active transport mechanisms require some energy source: you're right, they use the ATP.
Exocytosis and Endocytosis
Because the phospholipid molecules can move around with relative ease, the bilayer is flexible, self-sealing and fusible. It can form pockets which break off as sacks, carrying contents out (exo = out, cytosis = cell) of the cell. It can merge with a sack, then open it up, dumping the contents into the sack into the cell (endocytosis, endo = in). The actual mechanisms vary, depending on the type of cell (animals and plants use different endocytosis mechanisms), and the type of material being transported.
Keeping track of the types of transport can be a little confusing. Transport mechanisms can be classified by whether or not they use energy (active/passive) or whether or not they require molecules in the membrane (mediated/non-mediated). Notice that we can have carrier mediated passive transport mechanisms!
| Endocytosis | Phagocytosis (engulfs whole food particles) | Receptor Mediated | Internalizes molecules able to bind to specific resceptors in the membrane. |
| Carrier Mediated | Active transport:
primary (chemical energy) secondary (electrochemical gradient) | ||
| Passive transport: facilitated diffusion | |||
| Passive Transport (no protein in membrane required) | Diffusion filtration osmosis | ||
| Pinocytosis (engulfs dissolved molecules in liquid) | Internalizes any molecule already dissolved in liquid | ||
Web reading
For more details, check out narrated, animated tutorial on receptor-mediated transport at Freeman's site.
© 2005 - 2025 This course is offered through Scholars Online, a non-profit organization supporting classical Christian education through online courses. Permission to copy course content (lessons and labs) for personal study is granted to students currently or formerly enrolled in the course through Scholars Online. Reproduction for any other purpose, without the express written consent of the author, is prohibited.